|
Rutherfordium - Elementary my dear Ernest.
John Campbell
(A version of this was first written for newspapers throughout New Zealand. Another was
published in the New Zealand Science Teacher 86 36-7 1997.)
After years of
international haggling and horse trading, Ernest Rutherford has finally and formally
become the only New Zealander to have a chemical element named in his honour.
First a little background in chemistry and physics.
Our world and extensive galaxies comprise just 92 chemical elements. Of these only 81 are
truely stable. Those heavier than bismuth are radioactively decaying away until eventually
there will be none left. Those existing naturally, such as uranium, do so solely because
they are decaying at such a slow rate that there are still significant numbers of these
atoms remaining today, some 15 billion years since the elements first formed.
For a hundred years
we have known of the existence of the electron, the first object to be discovered which
was smaller than an atom. For eighty years we have known that the chemistry of an atom is
governed by the number of electrons in orbit about an atom. Just prior to that it was
Ernest Rutherford who showed the atom was a nuclear entity, with almost all its mass in a
nucleus less than a thousandth the diameter of an atom. (If the orbital electrons of the
atoms making up our body were pushed into the nucleus, as happens in a neutron star, our
body would fit into a small grain of sand.)
The nucleus
consists of protons (the nucleus of a hydrogen atom) and neutrons. Ernest Rutherford was
the first person to propose that the neutron had to exist and one of his colleagues
discovered it. Neutrons are electrically uncharged and, if isolated outside the nucleus,
decay after eleven minutes into a proton and an electron. Atoms have as many protons in
their nucleus as they have electrons in orbit. Hence the chemistry of an element is
effectively determined by the number of protons in its nucleus, the atomic number, which,
for those known to occur in nature, ranges from 1 for hydrogen to 92 for uranium.
back to top
Beyond Uranium
The heavy elements just above uranium were made in nuclear reactors where neutrons entered
a nucleus and the resultant heavier nucleus decayed by beta decay, thus moving one higher
in the periodic table. To the discoverer went the honour of proposing a name for each new
element. Neptunium (Np, A = 93) and plutonium (Pu, A = 94) were natural successors to
uranium. Names such as americium (Am, A = 95), berkelium (Bk A = 97) and californium (Cf,
A = 98) commemerated the place of discovery. Curium (Cm, A = 96), einsteinium (Es, A =
99), fermium (Fm, A = 100), mendelevium (Md, A = 101) and lawrencium (Lr, A = 103)
honoured the discoverers' scientific heroes. Nobelium (Nb, A = 102), claimed first, but
never substantiated, by an international group working at the Nobel Institute in
Stockholm, was named in honour of the man who left his fortune for prizes to promote
science.
Fermium (A = 100)
was the heaviest element obtainable from reactors. Those beyond fermium became special.
The production of
these heavy nuclei became more important when, between 1966 and 1972, new theories of the
nucleus predicted that the arrangement of particles in nuclei of atomic number around 114
should again be stable. But where were these elements? Renewed extensive searches of
nature still failed to find them.
They could only be
made by accelerating nuclei of around ten to twenty protons and neutrons to high speeds in
large accelerators and smashing them into the heaviest nuclei abundently produced in the
reactors.
The heavier the
element the harder its manufacture become and the shorter time it survived, until only
three laboratories specialised in this work: Berkeley in California, Darmstadt in Germany
and Dubna in Russia. And from element 102 on, the controversies started. These were fueled
by the cold war of the time.
back to top
Element 104
In 1964 the Dubna group, led by G N Flerov, claimed to have manufactured one isotope of
element 104 by smashing neon nuclei into plutonium. They proposed the name kurchatovium
(Ku), in honour of the Soviet nuclear physicist Igor Kurchatov.
Albert Ghiorso and
co-workers at the Lawrence Berkeley Laboratory of the University of California spent a
year attempting to repeat this work but finally had to conclude that element 104 could not
have been manufactured by Dubna. In 1969 the Berkeley team produced element 104 in an
entirely different way. They bombarded the world's supply of californium with high speed
nuclei of carbon atoms.
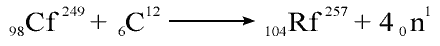
The isotope of
element 104 thus produced survived typically less than four seconds so could be uniquely
identified by its half-life and the energy of the alpha particles it emitted in the
process of spontaneous radioactive decay.
In November of
1969, at celebrations marking the centennial of Mendeleev, the father of the periodic
table, Al Ghiorso proposed that element 104 be named rutherfordium (Rf) because Ernest
Rutherford was one of his heroes. ``We are suggesting that element 104 be called
rutherfordium, after Lord Rutherford, the great pioneer of nuclear science. If in the
course of further experiments, contrary to our present expectations, we do confirm the
earlier findings of the Dubna group of approximately three-tenths of a second
spontaneous-fission activity, we will withdraw our suggested name and accept that proposed
by the Soviet group, kurchatovium.''
back to top
This was most
fitting as it was Rutherford who had first explained the nature of radioactivity, that one
element was decaying into another. That concept had been such an advance in science that
Rutherford had been awarded the 1908 Nobel Prize in Chemistry. Also he named the alpha
particles.
For nearly two
decades the world lived with three names for element 104. Each country used its own name,
the Oxford Dictionary listed both but politically correct periodic tables used an interim
name Unnilquadlium, the latin for one zero four, or Unq for short.
To solve the
impass, a Transfermium Working Group, a joint committee of the International Union of Pure
and Applied Physics and the International Union of Pure and Applied Chemistry, was set up
in 1985 to determine precedence of discovery for all elements beyond fermium. This would
allow unique names to be assigned. In 1992 the committee concluded that the two groups
should share credit for discovery of the elements 104 and 105. This conclusion was
bitterly rejected by the Berkeley group and others.
An August 1994
meeting adopted a new rule that no element could be named after a living person. Since
both Albert Einstein and Enrico Fermi had been alive when they had had element names
proposed in their honour, this move was a ploy to take the very much alive Glenn Seaborg's
name off element 106 so that that element could then be renamed rutherfordium. This left
element 104 open to be be renamed dubnium. Confusion reigned. Elements 104 to 109 were to
be named but international arguments continued over most of these. As I had already set an
exam question for 1994 based on element 104 being named rutherfordium I supported the
Americans.
back to top
In 1997 a final
compromise was reached and all phases of naming were passed. 104 rutherfordium (Rf), 105
dubnium (Db), 106 seaborgium (Sg), 107 bohrium (Bh), 108 hassium (Hs) and 109 meitnerium
(Mt). There are still problems. Bohrium, rather than the proposed nielsbohrium, has the
same name as the element boron (borii) in both Russian and German. And the American group
probably wont, in practice, accept dubnium in place of hahnium which they had proposed in
honour of Otto Hahn.
It is significant that both Niels Bohr and Otto Hahn first became internationally famous
while working with Ernest Rutherford.
The suggestion has
been made that future names be picked jointly by the Americans, the Germans and the
Russians who work in the field. When the other two groups have repeated the work the
discoverer will be asked to suggest a name which is satisfactory to all three. Only then
will it go to the formal naming committee.
The naming of
elements 110 to 112, all recently discovered, is being held over. Even so, the holy grail
of stable heavy-nuclei still eludes their creators.
But our Ern has his element.
From its position
in the periodic table, rutherfordium should have similar chemistry to hafnium. Its longest
living isotope has a half-life of about 70 seconds. Only a few thousand atoms of
rutherfordium have ever been manufactured and probably no more than 100 of these atoms
have ever been chemically isolated using a special cation exchange column. As you read
this there will most likely be not one atom of rutherfordium in existence, unless one of
the three groups are painstakingly manufacturing it for further experiments.
Fame is but
fleeting. However it is too much of a gamble to hold back a favourite name on the chance
that stable heavy-nuclei will eventually be manufactured.
back to top
References
I am most grateful for many discussions with Al Ghiorso concerning events mentioned in
this article.
Rutherford
Scientist Supreme
John Campbell, AAS Publications, 1999
contact: john.campbell [at) canterbury.ac.nz
The Elements Beyond Uranium
Glenn Seaborg and Walter Loveland, John Wiley and Sons, 1990.
The Discovery of Elements 95-106
Al Ghiorso, in the Welch Conference Proceedings
Fifty Years with the Transuranium Elements, Oct 22-23, 1990.
A History and Analysis of the Discovery of Elements 104 and 105
Hyde, Hoffman and Keller, Radiochimica Acta 42 57-102 1987.
back to top
Appendix - Recent Work Towards Stable Heavy Elements.
As the researchers
pushed towards higher elements the yields got smaller and the half-life of the elements
became shorter, ie they were less stable than their predecessors. For example, in 1996 the
Darmstadt group produced element 112 with mass number 277 which had a half-life of only
280 microseconds.
In December of 1998
the Russian group completed a joint experiment in which they had bombarded Livermore's
plutonium-244 sample with calcium-48 ions accelerated in a heavy ion cyclotron. The
bombardment was maintained for 40 days during which they produced just one atom of element
114. This work was reported in the Oct 18 1999 issue of Physical Review Letters.
What was exciting is that that isotope had a half-life 100,000 times longer than that of
element 112, the last new element found before element 114. Throughout 1999 the experiment
was repeated twice with the discovery of another isotope whose half-life was one second.
Two other isotopes were discovered independently by the Lawrence Berkeley team and another
Dubna team.
Are they
approaching the island of stability? Looks like it. The long lived isotope re-invigorated
heavy element research at Lawrence Berkeley National Laboratory where by the end of 1999
they had discovered elements 116 and 118. One problem is how does one know when a stable
element has been produced in quantities of just one or two atoms? It was Rutherford who
proclaimed radioactivity as by far and away the most sensitve method of detection of
atoms. So probably we will have to await the discovery of even higher elements whose
half-lives first increase then a gap to those whose half-lives once again decrease with
element number.
Watch this space.
back to top
|

